 In this Review, we summarize the underlying molecular mechanisms of physiological and pathological hypertrophy, with a particular emphasis on the role of metabolic remodelling in both forms of cardiac hypertrophy, and we discuss how the current knowledge on cardiac hypertrophy can be applied to develop novel therapeutic strategies to prevent or reverse pathological hypertrophy. In the past decade, a growing number of studies have suggested that previously unrecognized mechanisms, including cellular metabolism, proliferation, non-coding RNAs, immune responses, translational regulation, and epigenetic modifications, positively or negatively regulate cardiac hypertrophy. Monsoro-Burq AH, Duprez D, Watanabe Y, Bontoux M, Vincent C, Brickell P, Le Douarin N. Each form of hypertrophy is regulated by distinct cellular signalling pathways. Molecular distinction between physiological and pathological cardiac hypertrophy: experimental findings and therapeutic strategies. Hypertrophy initially develops as an adaptive response to physiological and pathological stimuli, but pathological hypertrophy generally progresses to heart failure. ‘Pathological’ cardiac hypertrophy is a condition that is characterized by the thickening of the heart muscle, a decrease in the size of the chambers of the heart, and a reduced capacity of the heart to pump blood to the tissues and organs around the body. 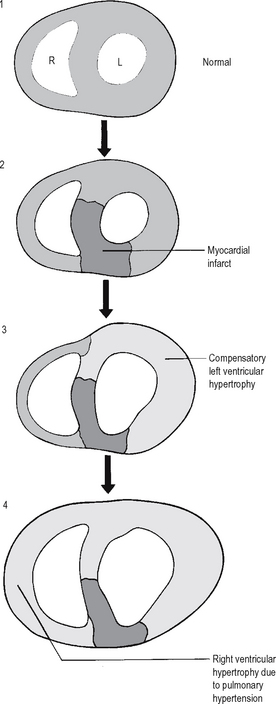
There are two types of hypertrophy: physiological and pathological. When contractile performance is perturbed or reduced in response to diverse (patho-)physiologic stimuli, the heart typically remodels and hypertrophies, in association with increases in myocyte cell volume (). Therefore, in the adult heart, instead of an increase in cardiomyocyte number, individual cardiomyocytes increase in size, and the heart develops hypertrophy to reduce ventricular wall stress and maintain function and efficiency in response to an increased workload. The primary function of the heart is to contract and pump blood. Cardiovascular function can decline in old age, and the disease independent factors that induce these changes are not well known. The mechanism by which these channels induce pathological hypertrophy is unclear, at least to us, but is thought to involve Ca 2+ influx through the TRP channels810. Cardiomyocytes exit the cell cycle and become terminally differentiated soon after birth. TRP channel expression is increased in hearts with pathological stress and their presence has been linked to the induction and maintenance of pathological hypertrophy8, 64. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
